

New Zealand Bee Pollen Database
Pollen morphology and descriptive features
Introduction
When shed from a flower or conifer pollen cone, a pollen grain is an independent organism, albeit short-lived. Within an outer protective wall the grain contains protoplasm and 2 or 3 nuclei, as well as food supplies in the form of starch granules. If the grain arrives on the receptive stigma of a compatible flower or the equivalent location in a conifer seed cone, it germinates to form a pollen tube down which nuclei pass to fuse with nuclei in the flower or conifer cone ovary and produce a seed.
Details of pollination, including the morphology of pollen grains, are consistent within individual species, families, and higher orders of plants, presumably because they are critical to the survival of a species. So, for example, the size and shape of pollen of manuka (Leptospermum scoparium complex) varies within narrow limits no matter the size of the parent plant or which part of the country it derives from. Manuka pollen is quite similar but recognisably different to the smaller and more compact pollen of kanuka (Kunzea species), but both share features such as flattened shape and united furrows with other members of the myrtle family (Myrtaceae), which includes rata and pohutukawa (Metrosideros species), ramarama (Lophomyrtus bullata), and eucalypts. Other plant families have their own characteristic pollen, some being very uniform among different genera, others less so. This means that an unknown pollen grain may frequently be identified to family, often to genus, but only sometimes to species, although if it is known to originate from a limited geographic area this may greatly restrict the range of possibilities.
For identification using a light microscope, we are mainly involved with details of the pollen grain wall, although the presence of abundant starch granules in the protoplasm, and the colour of pollen in fresh bee loads are also features that can be used in some cases. The wall consists of two main layers, the intine, an inner layer which encloses the protoplasm and which does not stain in our preparations, and the exine, an outer layer which does take stain and which displays characteristic internal and surface structural features. The intine is generally thin but may be thickened under apertures; along with the protoplasm it is quickly destroyed by acetolysis treatment. The exine is formed from a durable polymer, sporopollenin, which resists decay and only darkens gradually with acetolysis treatment.
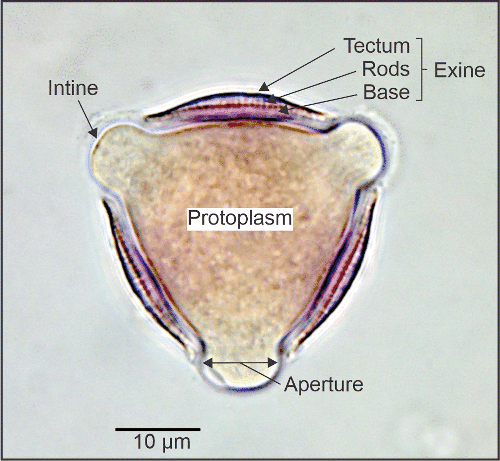
Pollen grain structure: an optical section of Pittosporum crassifolium pollen.
|
Because the exine walls of pollen grains are preserved in peats and other sediments, identification of its parent plants has been the subject of intense study by paleontologists engaged in climate and vegetation history. Pollen morphology is also very relevant to the study of plant evolution. These endeavours have resulted in numerous publications surveying the pollen flora of plant groups and geographic regions, and an extensive terminology for details of exine structure, mostly derived from analysis of acetolysed material (a good glossary is provided by Punt et al., 2007). The descriptions of morphological features used here are based on a scheme devised by the British melissopalynologist Sawyer (1981, 1988, with some modifications. This is designed for use with unacetolysed pollen and uses a simplified terminology.
The exine in flowering plants typically has several constituent sub-layers, although the upper ones may be absent in some species:
- a) a base layer, continuous and more or less unstructured, almost always present;
- b) a layer of rods (columellae), perpendicular to the base layer;
- c) a tectum, or roof supported on the rods: this may be continuous, or perforated, or may exist only as a mesh connecting the tops of rods;
- d) ornamentation comprising spines, warts, elongate ridges, or other elements arranged randomly or in a regular pattern on top of the tectum.
Where present, the arrangement of rods, tectum and ornamentation, along with systematic thickness variations, provide features for recognition of particular species.
Apertures are formed by the regular thinning of the exine, either in elongated areas called furrows or colpi, or of more limited extent, called pores. These serve as exit points for germination of the pollen, and also allow expansion and contraction of the protoplasm according to its degree of hydration. Often, furrows have pores in their mid-points, developed in a different (lower) level of the exine.
The regions of a pollen grain are described using terms applied to the Earth. In dicotyledonous flowering plants, apertures are commonly arranged at equidistant meridians, with pores (if present) at the equator. In some species, pores or furrows are scattered evenly over the whole surface. An equatorial view looks perpendicularly upon the equator of the pollen grain, while a polar view is one centred on the polar region. In monocotyledonous flowering plants (grasses, lilies, palms, etc.) there is usually only one aperture (pore or furrow), which is located at a pole.
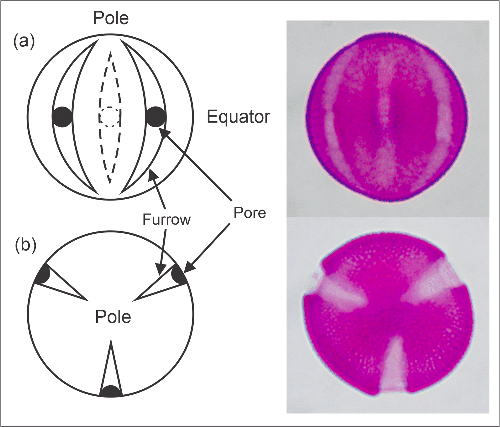
Morphological regions of a pollen grain with 3 furrows and pores:
(a) equatorial view, (b) polar view.
At right, Alectryon excelsus pollen grains in equivalent views.
|
Key features for identification
The online key enables the user to home in on a possible identification by entering observed features one at a time (and in any order). At each step, a range of best matches is displayed, and detail pages with illustrations and full descriptions can be brought forward. Each page also has botanical classification, plant common names, and remarks on the degree of discrimination which can be achieved among similar or related species. Where a number of species cannot easily be distinguished by their pollen morphology, a pollen type name is recommended. The images of pollen have been taken at high magnification (using a 100x oil immersion objective), usually of both acetolysed and unacetolysed material.
In the key, alternative interpretations or variations are included. For example, a species usually with 3 furrows but reasonably frequently with 4 will be recovered as a possible match by specifying either 3 or 46 furrows. It can be difficult, and perhaps unprofitable, to identify single or rare pollen specimens. Try to gain an understanding of an unknown pollen types features by examining several specimens in different orientations.
1. Aggregation
Most pollen is released as single units. Pine and podocarp pollen (e.g., Monterey pine, Pinus radiata; and kahikatea, Dacrycarpus dacrydioides) may appear to comprise aggregates of grains, but only the central body contains the living protoplasm. Two or three air sacs are attached to this: these have a spongy internal structure, the character of which may itself be diagnostic for different species. In a few flowering plant families, pollen is typically produced as multiple units, the morphology of the individual grains being affected by this close adherence. For example, groups of 4 pollen (tetrads) in tetrahedral arrangement are characteristic of the heath family (Ericaceae). The grains of the tetrad may be equal in size (Erica, Gaultheria) or some may be reduced in size leaving only one functional grain (Leucopogon). Tetrads with equal-sized grains arranged in one plane occur in kaiwhiri (Hedycarya arborea). Pollen of wattles (Acacia spp.) is released in polyads, groups of many individual grains. This initial key feature separates the following categories:
- 1.1. Single pollen.
- 1.2. Single pollen with air sacs.
- 1.3. Pollen united in groups of 4.
- 1.4. Pollen united in groups of more than 4.
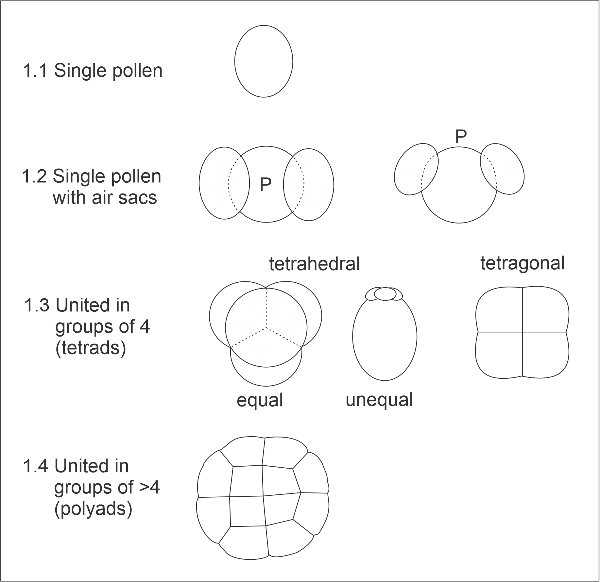
Diagrams of basic pollen forms (P = position of pole).
|
2. Size
This is the maximum dimension measured using an eyepiece graticule, as described in a previous section. The dimension should include any protruding spines or other surface ornamentation (but not protruding protoplasm). For tetrads and polyads, measure the overall dimension of the aggregate. Size classes are:
- 2.1. Very small, less than 20 µm. This category includes a number of taxa common in New Zealand honeys; for example, kamahi (Weinmannia racemosa), hinau (Elaeocarpus dentatus), trefoils (Lotus spp.), manuka (Leptospermum scoparium s.l.), and kanuka (Kunzea spp.). The tiny pollen of kawakawa (Piper excelsum) is not uncommon in bee loads from scrub and forest areas.
- 2.2. Small, 2030 µm.
- 2.3. Medium, 3050 µm. Small and medium sized pollen grains form the bulk of other species seen in bee pollen loads and in honeys, for example various legumes including white clover (Trifolium repens).
- 2.4. Large, 50100 µm.
- 2.5. Very large, more than 100 µm. Large and very large pollen grains are relatively uncommon in honeys. In bee loads, pollen of pine (e.g., Pinus radiata) and the various podocarps fall into these size classes.
3. Shape in equatorial view
This is the general shape of the outline of the pollen, as seen with the equator crossing the grain approximately midway. The categories are:
- 3.1. Round or irregularly round. Grains in which the polar dimension is more or less equal to the maximum equatorial dimension, producing a circular or more or less equidimensional outline.
- 3.2. Flattened oval. Grains in which the polar dimension is shorter than the maximum equatorial dimension; for example, most Myrtaceae pollen. The 3-dimensional shape is said to be oblate. Very flattened grains tend to lie on a flat side, so one in equatorial view may be difficult to find. The boat-shaped pollen of palms, cabbage trees and related plants, which have a single furrow located at one pole, are also included here.
- 3.3. Elongate. Grains in which the polar dimension is longer than the maximum equatorial dimension (prolate).
- 3.4. Very elongate. Grains in which the polar dimension is much longer (>1.5x) than the maximum equatorial dimension (perprolate).
- 3.5. Different at each pole. The two polar halves of the pollen differ. This includes some species which have a single (polar) aperture and which may also be scored as round or flattened, as well as a few unusual multi-aperture species which have grains expanded at only one pole; e.g., vipers bugloss (Echium vulgare). Saccate conifer pollen are also scored here because the air sacs are offset towards one pole.
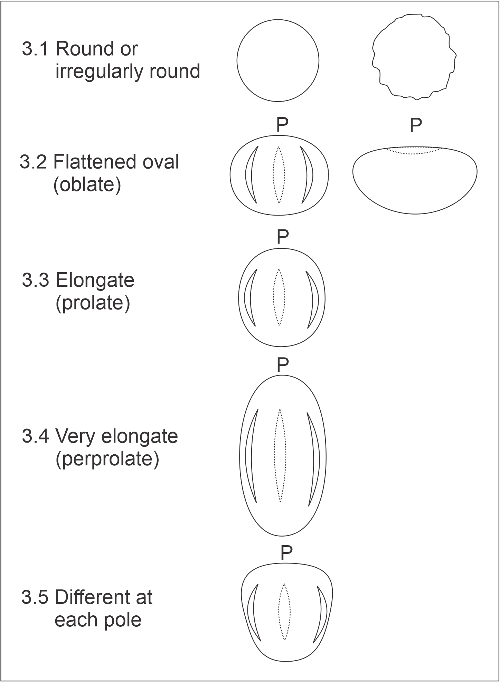
Equatorial view shape categories (P = position of pole).
|
4. Shape in polar view
This is the general outline shape of the pollen, as seen looking down on one pole. The arrangement of apertures commonly affects the outline. Prolate and especially perprolate pollen grains tend to lie on their side, so polar views of these species may be difficult to find. Categories are:
- 4.1. Round or irregularly round. For example, mahoe (Melicytus ramiflorus), and poplar (Populus spp.).
- 4.2. Triangular or rounded triangular (apertures at corners). This is a common shape for pollen with 3 apertures (e.g., rewarewa, Knightia excelsa), but also applies to the triangular pollen of harakeke (New Zealand flax, Phormium tenax) with its polar, branched furrow.
- 4.3. Triangular or lobed (apertures in sides). Examples are kakaramu (Coprosma lucida), rounded triangular with apertures in sides, and wineberry (Aristotelia fruticosa), with trilobed pollen.
- 4.4. Oval. This category includes mainly pollen which have a single furrow located at one pole; for example, ti pore (Cordyline fruticosa), and hutu (Ascarina lucida).
- 4.5. Multisided, multilobed, or irregular. Pollen with more than 3 meridional furrows often fall into this category; for example, the multisided tawari (Ixerba brexioides), and round to multilobed borage (Borago officinalis). The 6-sided outline of dandelion (Taraxacum officinale) and related plants, caused by prominent spiny ridges, also places these species in this category.
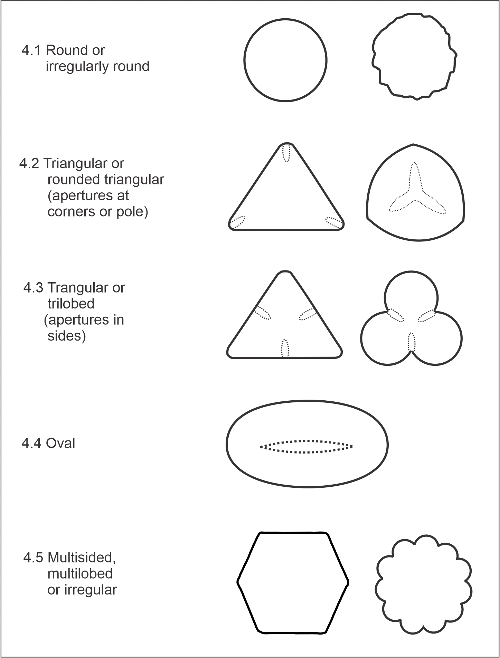
Polar view shape categories (P = position of pole).
|
5. Number of apertures
Furrows containing pores are counted as one aperture. In compound grains (tetrads or polyads), count the number for a single subunit. The individual grains of regular Ericaceae tetrads generally have 3 apertures (combined furrows with pores), but those of irregular tetrads (some Leucopogon species) may have more. The individual grains of Acacia polyads display a single rectangular to circular furrow. The key separates the following categories:
- 5.1. None or indefinite apertures. Examples are poplar (Populus spp.), and various conifers, including Monterey cypress (Cupresses macrocarpa), the pollen of which splits irregularly.
- 5.2. One aperture. Mostly monocots; e.g., grasses, palms (e.g., nikau, Rhopalostylis sapida), etc., but also a few dicots (e.g., hutu, Ascarina lucida).
- 5.3. Two apertures. Uncommon, but includes the two-aperture pollen of pukatea (Laurelia novaezealandiae, with furrows), Fuchsia (with pores), and karaka (Corynocarpus laevigatus, with combined pores and inconspicuous furrows).
- 5.4. Three apertures. The typical number for dicot flowering plants. Some which normally have 3 apertures may occasionally include specimens with 4 apertures. In such cases, both number classes are scored in the key.
- 5.5. Four to six apertures. Pollen with more than 3 apertures often displays a range of numbers. For example, red beech (Fuscospora fusca) pollen may have from 5 to 7 or more apertures.
- 5.6. Seven to twelve apertures. These may be arranged meridionally around the equator of the pollen (e.g., borage, Borago officinalis, with about 10 meridional furrows and an encircling equatorial furrow), or scattered over the grain (e.g., the pores of plantains, Plantago spp.).
- 5.7. More than twelve apertures. Relatively few taxa, typically those with many pores scattered equidistantly over the surface of the pollen; e.g., chickweed (Stellaria spp.).
6. Aperture types
These are illustrated in the Introduction.
- 6.1. Pores only. Examples are kiekie (Freycinetia banksii), with a single pore, and rewarewa (Knightia excelsa), with 3 pores.
- 6.2. Furrows only. Examples are canola (Brassica napus), with 3 furrows, and thyme (Thymus vulgaris), with 6 furrows.
- 6.3. Furrows with pores. Very common in dicot flowering plants; for example, matagouri (Discaria toumatou), with 3 combined furrows and pores, and kumarahou (Quintinia serrata), usually with 5 furrows and inconspicuous pores.
- 6.4. United or irregular furrows. Myrtaceae pollen typically has 3 (rarely 4) furrows joining together at the poles (a condition termed syncolpate)there may be a triangular island area at the pole (e.g., rata, Metrosideros robusta), or the furrows may unite in a point (manuka, Leptospermum scoparium). Irregular and united furrows occur in barberry (Berberis darwinii).
7. Surface pattern
Surface textures of pollen grains are best seen at high magnification, using 40x or a 100x oil immersion objective and focussing slowly down on to the top of the pollen. Each of the categories includes a range of variations.
- 7.1. Smooth or indefinite. With no definite pattern e.g., tawari (Ixerba brexioides).
- 7.2. Granular. With granules, warts, or irregular elevations e.g., bidibid (Acaena profundeincisa), and ling heather (Calluna vulgaris).
- 7.3. Striate. Thin, low parallel ridges, either meridional or in patches at different angles, sometimes branched. Generally faint and difficult to discern: in New Zealand examples are mostly confined to some Rosaceae (e.g., blackberry, Rubus fruticosus), and puka (Griselinia littoralis).
- 7.4. Net or pitted. A very common pollen surface pattern among the flowering plants, this may range from very fine with mesh openings less than 1 µm (e.g., rosemary, Rosmarinus officinalis) to coarse with mesh up to several microns across (e.g., wharariki, Phormium cookianum). The mesh size may vary systematically; e.g., finer at the poles or next to furrows, and the mesh walls may appear beaded (due to large supporting rods). Also included in this category are pitted surfaces.
- 7.5. Isolated dots due to spines or other projections. These may occur alone (e.g., coastal astelia, A. fragrans), or superimposed on a background of another surface pattern (e.g., buttercups, Ranunculus spp., with tiny spines on a granular to smooth background; and akiraho, Olearia paniculata, with broad-based spines on a pitted background).
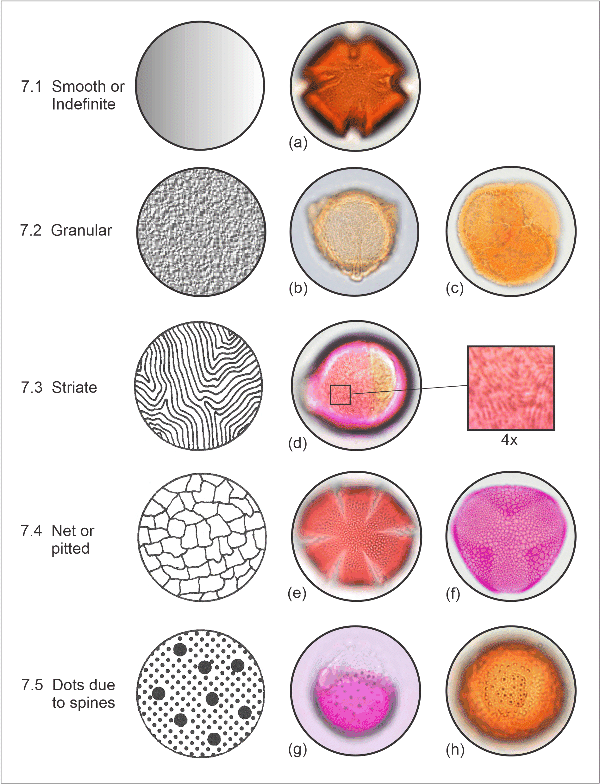
Surface pattern categories. (a) tawari, (Ixerba brexioides);
(b) bidibid, Acaena profundeincisa; (c) ling heather, Calluna vulgaris;
(d) peach, Prunus persica, with detail magnification;
(e) rosemary, Rosmarinus officinalis; (f) wharariki, Phormium cookianum;
(g) coastal astelia, Astelia fragrans; (h) buttercup, Ranunculus sp.,
with tiny spines
on a granular to smooth background.
|
8. Exine section
This feature describes the exine as seen in optical section: both the thickness and internal structure, and surface projections such as spines. These two aspects may both be scored.
- 8.1. Thin to medium, no visible rods. One or two stained exine layers without a clear rod layer; e.g., blackberry (Rubus fruticosus).
- 8.2. Medium with spaced rods, or beaded. Two stained layers with a clear row of regular, well-spaced rods between them (sheeps sorrel, Rumex acetosella). The outer layer may be discontinuous, giving a beaded appearance (in surface view the exine may have a net or pitted appearance); e.g., pukatea (Laurelia novaezelandiae).
- 8.3. Medium or thick with coarse external rods. Large rods form the outer layer; e.g., cranebill (Geranium molle).
- 8.4. Layer of close, thin rods. Exine thin to thick, but with thin, densely packed rods, which may be branched; e.g., silverbush (Convolvulus cneorum).
- 8.5. Long, thin spines. Large spines which are taller than the width of the base; e.g., houhere (Hoheria sexstylosa).
- 8.6. Large, broad-based spines. Spines which are no taller than the width of the base; e.g., nodding thistle (Carduus nutans).
- 8.7. Very small spines or projections. Spines or other discrete projections which are less than 1 µm in width; e.g., Himalaya honeysuckle (Leycesteria formosa).
- 8.8. Other projections. Projections variously club-shaped (hutu, Ascarina lucida), spiny ridges (hawksbeard, Crepis vesicaria), large warts (ling heather, Calluna vulgaris), etc.

Exine section categories.
|
9. Other features
These include aspects of pollen apertures, and of the intine and protoplasm:
- 9.1. Aperture edges thickened or projecting. Thicker exine may form a narrow band around pores (grasses; e.g., Lolium perenne) or furrows (whauwhaupaku/five-finger, Pseudopanax arboreus), or there may be a more complex, chamber-like arrangement at pores (kotukutuku, Fuchsia excorticata).
- 9.2. Cap or streak on apertures. Mostly, pores and furrows include only thinned exine, but sometimes there may be a central region of thicker tissue (plantain, Plantago lanceolata).
- 9.3. Granules or other projections scattered on apertures. Rather than a continuous cap, there may be granules or spinules. These are seen very clearly in Convolvulus (e.g., C. cneorum), and like aperture caps, may be shed as the protoplasm expands.
- 9.4. Intine swollen beneath apertures. This results in clear masses beneath the apertures (e.g., walnut, Juglans regia), but must be distinguished from contracted protoplasm leaving air spaces.
- 9.5. Cell contents granular. The appearance of the pollen protoplasm varies with age and species. A few species consistently have dense starch granules (e.g., buckwheat, Fagopyron esculentum).
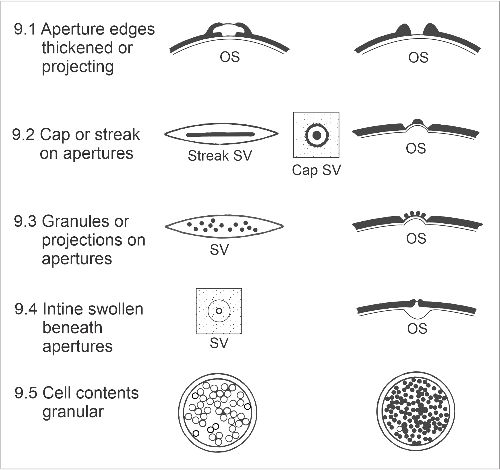
Other features (OS = optical section, SV = surface view).
|
10. Pollen pellet colour
Fresh dry bulk pollen in bee loads from many plants is a shade of yellow to brown and these are difficult to reliably identify on colour alone. However, some is distinctive; for example, the violet pollen of Phacelia (Newstrom-Lloyd et al., 2012), the red of horse chestnut (Aesculus hippocastanum), or the blue of Fuchsia. The colour can change with age of the pollen pellet, gradually becoming browner as the pollen decays. Where known, either from our own research or from Kirk (2006), Sawyer (1981) or Walsh (1978), the fresh bee load colour has been classed in a basic scale:
- 10.1. White
- 10.2. Grey
- 10.3. Brown or beige
- 10.4. Light yellow
- 10.5. Yellow
- 10.6. Orange
- 10.7. Pink or red
- 10.8. Violet or purple
- 10.9. Green
- 10.10. Blue to black
Cite this publication as:
"J.I. Raine, X. Li, L. Newstrom-Lloyd (2022).
New Zealand bee pollen database. "
© GNS Science, 2022










